Disclosures: Allen Beuershausen is the Co-founder of Sanitis and Harry Jarbath is the Founder of Sanitis. Sanitis is a global sales and marketing company distributing “next generation” infection control solutions for the health care, food processing and food service, janitorial, and transportation industries.
Abstract
Nosocomial infections are a serious and growing problem in many healthcare institutions in spite of great and frequently expensive efforts to bring them under control. There are multiple, interrelated issues that make it difficult to significantly reduce the spread of pathogens in health care institutions including inadequate assessments of threats, outdated hygiene protocols, poor compliance with existing protocols, and infection control and disinfection products that are either inadequate for their intended purpose or improperly applied. Fortunately, new technologies are available that can make it easier to reduce the incidence of these infections.
The authors of this paper work with many of these technologies on a daily basis and offer their insights into existing infection control products and systems as well as alternative technologies that have helped hospitals to reduce HAIs by 23% to 33%, and schools that have seen reductions in illness-related absences of up to 38.9%.
Main Article
Nosocomial infections, or HAIs (Hospital Acquired Infections), are the bane of health care facilities and their clients world wide, and the problem is not going away any time soon. Hand hygiene, environmental infection control, and improperly disinfected devices such as catheters are among the biggest contributors to the problem.1 Products and protocols that have been used for disinfection for many years are not doing the job even when infection control compliance is high, but a range of new eco-friendly disinfectants and devices might change that with wide spread adoption of proper protocols and strict compliance.
Disinfectants are now available that kill bacteria, viruses, and fungi without harming people or the environment2. UV lights can kill pathogens while rooms are idle, fogging systems can effectively disinfect entire rooms after contamination with the toughest pathogens, and electrostatic sprayers can provide virtually 100% coverage of surfaces with aqueous disinfectants in a fraction of the time and with a fraction of the chemicals that are necessary with traditional products and protocols.3 Novel hand hygiene products now control pathogens that traditional hand sanitizers cannot, and they provide persistence, i.e., continuous protection, to protect even when compliance rates are low.4,5
Protocols are Key
Proper disinfection and hygiene protocols that are strictly adhered to are critical in maintaining personal and environmental infection control. Even though health care facilities have those protocols in place, there are still gaps in the health care environment that allow pathogens like Clostridium difficile, MRSA, Staphylococcus aureus, and Acinetobacter baumannii, to spread. For example, it is very common for toilets in patient rooms to have no lids, thus allowing fecal bacteria to be ejected in an aerosol with each flush and contaminate surfaces throughout a room.1 Even with today’s efficient siphonic toilets, bacteria including Escherichia coli have been shown to remain viable in aerosol form for 4 – 6 hours.6 Another source of contamination are hospital water supplies (storage tanks and piping) and ventilation systems where harmful Aspergillus species are commonly found.7 For this reason, gap analysis to compare ideal versus actual operating conditions and food industry-style HAACP (Hazard Analysis Critical Control Point) plans can be extremely valuable for identifying all potential threats and bringing the environment and other sources of nosocomial infections back into compliance when problems do occur.
Soft surfaces such as curtains and upholstery often receive insufficient sanitizing and can harbor any number of pathogens. In fact, a 2011 study performed at one hospital in the University of Iowa hospitals and Clinics system showed that within 1 week of being laundered, 92% of hospital curtains were contaminated with pathogens, including methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant Enterococcus (VRE) species.8 Many broad-spectrum disinfectants used today can damage soft surfaces, so opportunities to eliminate pathogens on those surfaces are often missed.9 The use of disposable curtains is one way to help minimize the spread of disease, but there are less wasteful, more cost effective, and more environmentally friendly ways to manage those threats with appropriate disinfectants and application methods.
One of the easiest and most important means of reducing the spread of HAIs is to improve hand hygiene. Unfortunately, this can also be one of the most challenging problems to resolve because of antimicrobial preparations with limited efficacy and health care workers’ low compliance rates with existing protocols.10 Improved compliance in hand hygiene, with proper use of alcohol-based hand rubs, have been shown to reduce the nosocomial infection rate by as much as 40%.10 New, cutting edge antimicrobial hand hygiene products show great promise in reducing the transmission of disease through hand contact even further due to their greater broad-spectrum efficacy, their persistence, and their acceptance by workers who sometimes avoid sanitizing because of skin irritation.
Hand Hygiene
“An antiseptic product should be: broad-spectrum, fast-acting, and if possible, persistent.” 11
While this is an excellent recommendation, few if any hand sanitizers or surgical scrubs can provide the efficacy and persistence that the FDA recommends. Fortunately, that is changing with novel hand sanitizers based on more effective and innovative antimicrobial formulations, and new attention to monitoring for compliance with hand hygiene protocols is helping a great deal, as well. The transmission of HAIs through hand contact remains a huge problem. Healthcare facilities around the world use products with different active ingredients and different properties, and in light of the FDA’s recent ban on triclosan and their recently announced concern about other active ingredients in hand sanitizers, it is worth reconsidering the products being used.12 Table 1 provides an illustration of some of the more common active ingredients in hand sanitizers and illustrates the wide variation in their efficacy and toxicity. Furthermore, common products used in the home such as typical hand lotions can negate the antibacterial effects of active ingredients like CHG (chlorhexidine gluconate).13 It is likely that infection control professionals will see reason to switch to more effective soaps, rubs, and scrubs when more data is available to inform the infection control practitioners.
Table 1: Mechanism And Spectrum Of Activity Of Antiseptic Agents Commonly Used For Preoperative Skin Preparation And Surgical Scrubs 14
Source: Centers for Disease Control and Prevention, National Center for Emerging and Zoonotic Infectious Diseases, Division of Healthcare Quality Promotion
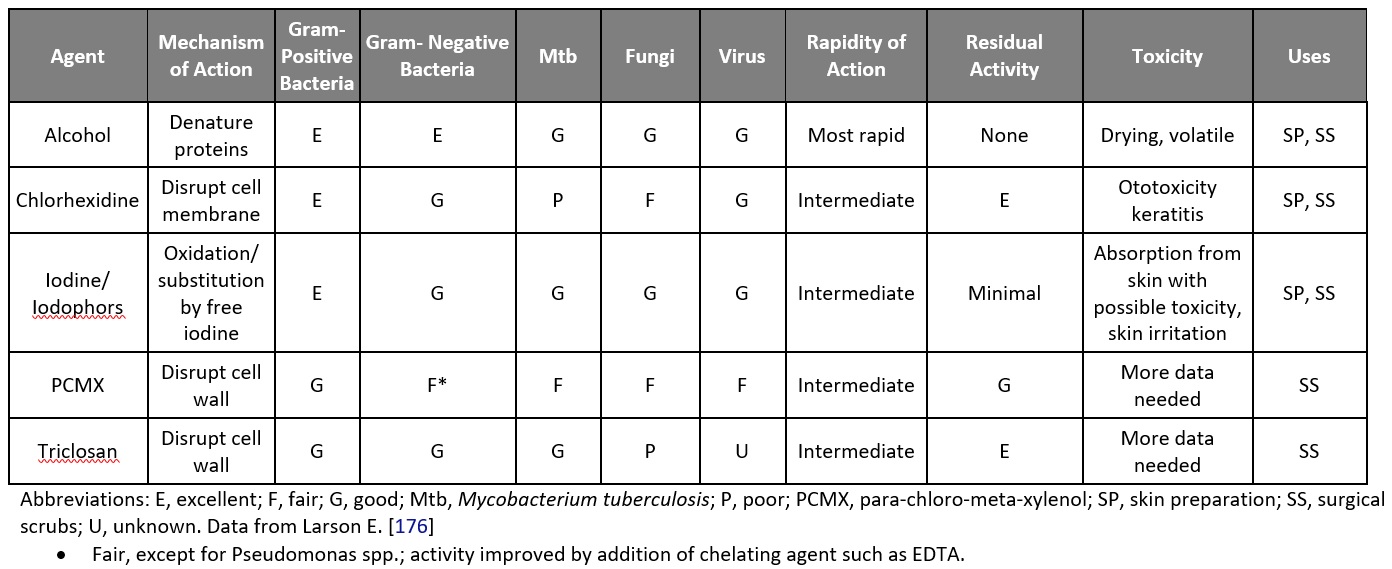
Alcohol-based hand sanitizers, or “rubs”, that are currently considered to be the “gold standard” of hand sanitizers because of their quick-acting effects on bacteria and enveloped viruses have little efficacy against non-enveloped viruses like norovirus, and have no persistence as illustrated in the CDC’s Table 1 above.15 Once they evaporate, which is generally within 15 seconds, skin can become re-contaminated immediately.15 Frequent use of alcohol-based rubs can lead to dry, cracked skin that harbors more bacteria than healthy skin, and compliance with hand hygiene protocols can fall off due to the discomfort caused by the use of traditional products.16 Many manufacturers of these traditional alcohol-based rubs are developing formulations that are less irritating to the skin, but they still lack persistence.
Chlorhexidine gluconate (CHG) is a common antibacterial and antifungal agent used in hand sanitizers and surgical scrubs in the health care industry, but it is not very effective against non-enveloped viruses.11 For this reason, CHG is often combined with alcohol to provide efficacy against viruses and increase its persistence. Some of the most popular surgical scrubs are formulated in this way. One advantage CHG does provide is persistence against bacteria and fungi for up to about four hours. Unfortunately, CHG can be very hard on skin.
Iodine/Iodophors have been used as antiseptics for the skin for many years, and they are effective against bacterial, viral, and fungal pathogens.11 Iodophors are used more commonly in hand sanitizing products, particularly as povidone-iodine which is relatively non-staining and less toxic and irritating as compared to iodine.11 While iodophors are bactericidal, virucidal, and fungicidal, most formulations require prolonged contact times in order to be effective.5
Chloroxylenol (PCMX) is seen in a number of antibacterial soaps and surgical scrubs, but unfortunately, it is one of the least effective antimicrobials, and the CDC requires a two minute wash time with PCMX in order to meet their minimum standards for hand hygiene.11 Most other hand hygiene preparations are effective within 15 to 30 seconds and they are more effective. PCMX works well against Gram-positive bacteria, and has fair efficacy against gram-negative bacteria, fungi, and certain viruses.11
Some of the newest antimicrobial soaps, rubs, and surgical scrubs contain the quatermonium compounds benzethonium chloride (BZK) or benzalkonium chloride (BAC).17 BZK and BAC have been used safely for decades as antiseptics and as preservatives in food, toothpaste, and other consumer products, and in many hand sanitizer formulations they provide broad-spectrum efficacy that is recommended by the FDA.17 Formulations including hand sanitizers, foaming soap, and surgical scrub combine BZK with synergistic compounds allow the products to provide up to six hours of persistence (Sanitis).4,5 These products have been demonstrated in clinical studies to provide up to six hours of persistence even through multiple hand washings, and long-term field trials in hospitals have shown dramatic reductions in HAIs in hospitals that have switched from traditional alcohol-based rubs to these products.18 An added benefit to BZK and BAC-based products is that skin irritation and dryness are generally avoided at the concentrations allowed by the FDA, and hand hygiene compliance is less of an issue since health care practitioners are less likely to avoid the products because of discomfort.
Disinfectants
Traditional disinfectants like bleach (sodium hypochlorite), quaternary ammonium cation compounds (“quats”), phenols, and peracetic acid (PAA) are effective and reliable disinfectants that have been used for many years to sanitize and disinfect hard surfaces in homes, offices, hospitals, and just about every place else.19 However, few of these can be used on soft surfaces such as bedding, upholstery, and curtains, and they all have shortcomings including toxicity, irritation of eyes and skin, flammability, corrosion or other damage of treated materials, and even a lack of broad spectrum efficacy.19
Fortunately, there are technologies in chemical disinfection that avoid the problems of traditional disinfectants such as acute toxicity, skin irritation, and corrosion of equipment. “Cutting edge” formulations with ingredients like chlorine dioxide, hypochlorous acid, and even the old standby, hydrogen peroxide, are providing equal or better results with true broad spectrum efficacy and without the serious hazards and limitations of their predecessors.19 These “next generation” disinfectants frequently have the added benefit of minimizing the possibility of giving rise to “super bugs”, i.e, bacteria that have evolved to be resistant to antibiotics and other disinfectants.
Chlorine Dioxide
Chlorine dioxide (ClO2) is an extremely effective “selective” oxidizer that has been used for many decades to sanitize municipal water systems around the world.20 In the past, it was usually only available as a gas that is generated on site. The gas itself can be hazardous if it leaks into the atmosphere, but in recent years manufacturers have developed ways to stabilize ClO2 in water and even to create small quantities of ClO2 solutions as needed. 21
Chlorine dioxide has a very different chemical structure and properties from bleach (sodium hypochlorite), and can be used much more safely in the concentrations used in hospital disinfectants. Innovations such as shelf-stable ClO2 products (Sanitis) provide broad spectrum antibacterial and antiviral efficacy and extremely effective odor elimination without the hazards associated with bleach and other disinfectants.22 One important benefit of chlorine dioxide is its ability to eliminate biofilms that can harbor pathogens such as Listeria monocytogenes and Aspergillus species. This makes it especially suitable for water treatment applications when dosed into hospital water systems at very low concentrations. Few disinfectants, including bleach, are capable of eliminating biofilms. ClO2 is very eco-friendly when used in solution, and it produces safer and fewer byproducts as a result of its antimicrobial activity than other disinfectants do.23
Hypochlorous Acid
Hypochlorous acid is a very effective antimicrobial that is recognized by the Centers for Disease Control (CDC) as having a more prolonged antibacterial effect and more free available chlorine than sodium hypochlorite while being potentially safer for the environment.11 Hypochlorous acid can be generated in a number of ways that include chemical precursors that are added to water or through “super oxidation” of water by specialized electrical equipment. According to the CDC, “Freshly generated superoxidized water is rapidly effective (<2 minutes) in achieving a 5-log10 reduction of pathogenic microorganisms (i.e., Mycobacterium tuberculosis, Mycobacterium chelonae, poliovirus, HIV, multidrug-resistant S. aureus, E. coli, Candida albicans, Enterococcus faecalis, Pseudomonas aeruginosa) in the absence of organic loading. However, the biocidal activity of this disinfectant decreased substantially in the presence of organic material (e.g., 5% horse serum).”19
One of the most interesting developments in hospital-grade disinfectants is the development of disinfectant precursor tablets that are dropped into water to create hypochlorous acid-based solutions as needed and at the strength the user desires. One manufacturer in particular has developed extremely cost competitive tablets that can generate an EPA-registered disinfectant that is National Sanitation Foundation (NSF) certified as a food contact surface sanitizer (classification D2, no rinse required) at low concentration, a hospital-grade disinfectant that kills bacteria, viruses, and fungi, including blood-borne pathogens and C. difficile spores at medium concentration; or a C. difficile disinfectant with a four minute kill claim at the product’s highest EPA registered concentration.24 Even with this broad spectrum efficacy, the product is considered to be non-hazardous under OSHA Occupational Health and Safety Standards on Toxic and Hazardous Substances number 29 CFR 1910.1200.24
As mentioned above, hypochlorous acid solutions can also be created by electrolyzing saline solutions of water on demand. The resulting “super oxidized” water is very effective as a disinfectant, and very safe for the environment.11 It can be generated at attractive costs, but the required equipment can be expensive in order to compensate for pH and other parameters, and it does require regular maintenance. The degree, difficulty, and cost of maintenance depend on the design of the equipment that varies from one manufacturer to another.
Hydrogen Peroxide Formulations
Hydrogen Peroxide (H2O2) has been used as a disinfectant for many years, and for good reason: it is a very effective broad-spectrum disinfectant. It is even sporicidal at higher concentrations (10 – 30% v/v).25 Unfortunately, it also has downsides as traditionally employed. Hydrogen peroxide decomposes quickly into water and oxygen, and it can be toxic at higher concentrations.25 The good news is that formulations now exist that get around some of the shortcomings of H2O2.
One of the most effective ways to use these new formulations is through the use of whole-room fogging systems. These systems create a fine mist of hydrogen peroxide-based disinfectant that makes its way into the difficult-to-reach areas. While this requires taking a room out of commission for up to three hours and sealing it to contain the mist in the target area, it can be a very effective way of eliminating even the most difficult pathogens including C. difficile.
Important Note about Disinfectants
Regardless of what disinfectant is used, it is vital that proper protocols (outlined by the manufacturer) for disinfection are in place and that they are strictly adhered to in order to achieve the desired result. These protocols must include cleaning surfaces before they are disinfected to ensure that the chemical of choice makes contact with any pathogens. The degree of disinfection by any product is based on both the chemistry and the contact time it has on the surface.
A far better way to apply disinfectants in an environment is through the use of electrostatic sprayers with disinfectants that do not need to be wiped off. Electrostatic sprayers impart an electrical charge to the droplets of disinfectant as they leave the nozzle of the sprayer.26 This electrical charge causes the droplets to be attracted to the target object and the droplets repel each other once they are on the surface. This electrical repulsion essentially causes the disinfectant to surround the object to ensure virtually 100% coverage in three dimensions, even covering surfaces that are hard to reach by hand or typical sprayers.25 Electrostatic sprayers drastically reduce the amount of time and chemical required to disinfect a room, and they are available in backpack, cart, and cordless handheld models. Electrostatic sprayers have been used for decades to apply pesticides to produce in the field and to apply paint to cars on assembly lines, so it is a highly proven technology.26
Ultraviolet Light
Ultraviolet lights are becoming more popular in health care facilities, and for good reason. Ultraviolet Germicidal Irradiation (UVGI) inactivates pathogens by targeting nucleic acids (DNA and RNA)27 Ultraviolet light is effective against bacteria, viruses, and fungi, and it has found widespread use in disinfecting surfaces, air, and drinking water.27 Ultraviolet systems are available as fixed lighting units, portable fixtures, and even robotic units that eliminate some of the biggest limitations of these systems. However, UV systems are not without their limitations.
Just as with chemical disinfectants, UV light systems are dependent on the intensity (concentration) of the light and the contact time.27 Microbes can only be killed if they are irradiated by the light, so the light source(s) must be arranged or moved to ensure that they are exposed to every surface with the appropriate intensity for the proper amount of time.28 This can be a challenge even for properly trained operators. Some hospitals have found that their rates of C. difficile infections have increased after spending upwards of $500,000 on UV equipment and hiring several technicians to operate it. Robotic systems can help with this by automating the process and reducing operator error. In any case, it is imperative that operators of UV systems are well-trained and vigilant in their implementation of manufacturer recommended protocols. Having said that, small UV cabinets have proven to be very effective in disinfecting medical devices where the process is easily controlled.
Conclusion
In spite of the challenge of minimizing nosocomial infections, technologies exist that can make it far easier and safer to reduce the spread of illnesses than many realize. The products that utilize these technologies, when used with appropriate care and protocols, can help hospitals and clinics to dramatically reduce the incidence of HAIs and the economic and human costs associated with them. Fortunately, when the ROI on these alternative products and systems is considered, they can pay for themselves very quickly.
Even with these new approaches in infection control, it is still incumbent upon infection control specialists to identify all potential sources of HAIs and address them systematically and holistically. Health care practitioners and custodial staff must remain vigilant and be held accountable for following the processes and procedures that are laid down to minimize the spread of illnesses. With this approach and the right infection control products, pathogens that have become endemic throughout health care systems worldwide may be controlled to a degree not seen before.
References
- Furness C, Wallace N (2015) Hand Hygiene Compliance Auditing Does Not Work. InfectionControl.tips 12:1-5 https://infectioncontrol.tips/2015/12/27/1291/
- Birko, “10 Reasons Why You Should Be Using Chlorine Dioxide”, Food Safety Magazine, February/March 2005.
- Whitely B (2016) Electrostatics: A Solution for Healthy Touch Point Surface Treatments. InfectionControl.tips. 5:1-3 https://infectioncontrol.tips/2016/05/10/electrostatics-solution/
- Bioscience Laboratories. (2014) Clinical Evaluation of the Persistent Antimicrobial Efficacy of Three Test Products and One Control Product Based on ASTM E2752-10. Accessed October 19, 2016. http://www.supplyurneeds.com/uploads/6/5/2/1/65218579/clinical_evaluation_of_the_persistent_antimicrobial_efficacy.pdf
- Haskins Lab (2012) Pace University. Accessed October 20, 2016. http://www.pace.edu/dyson/initiatives-research/haskins-laboratories
- Gerba CP, Wallis C, Melnick JL. (1975) Microbiological hazards of household toilets: droplet production and the fate of residual organisms, Appl Microbiol. 30:229–37
- Burge, H., & Aerobiologist, E. P. K. C. (2009). Sources and control of opportunistic fungi in the hospital environment. Environmental Reporter, 8(1).
- Brunk D (2011) Beware of Curtain! Pathogens Plentiful on Hospital Partitions”, American College of Emergency Physicians News, December. Accessed October 21, 2016 https://www.acep.org/content.aspx?id=82855
- Connell, B (2016) What’s Really Hiding behind Hospital Privacy Curtains? Medline Clinical Solutions. June. Accessed October 21, 2016 http://mkt.medline.com/advancing-blog/whats-really-hiding-behind-hospital-privacy-curtains/
- Dettenkofer, M., & Conrad, A. (2010). Hand Hygiene for the Prevention of Nosocomial Infections. Deutsches arzteblatt international, 107(8), 139-139.
- CDC Morbidity and Mortality Weekly Report, Guideline for Hand Hygiene in Health-Care Settings, October 25, 2002 / Vol. 51 / No. RR-16
- Food and Drug Administration (2016) FDA issues final rule on safety and effectiveness of antibacterial soaps. US FDA. Sept 2, 2016. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm517478.htm
- Frantz, S. W., Haines, K. A., Azar, C. G., Ward, J. I., Homan, S. M., & Roberts, R. B. (1997). Chlorhexidine gluconate (CHG) activity against clinical isolates of vancomycin-resistant Enterococcus faecium (VREF) and the effects of moisturizing agents on CHG residue accumulation on the skin. .Journal of Hospital Infection, 37(2), 157-164
- Mangram, A. J., Horan, T. C., Pearson, M. L., Silver, L. C., Jarvis, W. R., & Hospital Infection Control Practices Advisory Committee. (1999). Guideline for prevention of surgical site infection, 1999. American journal of infection control, 27(2), 97-134.
- Blaney, D. D., Daly, E. R., Kirkland, K. B., Tongren, J. E., Kelso, P. T., & Talbot, E. A. (2011). Use of alcohol-based hand sanitizers as a risk factor for norovirus outbreaks in long-term care facilities in northern New England: December 2006 to March 2007. Am J Infection Control, 39(4), 296-301.
- Klevens, R. M., Edwards, J. R., Richards Jr, C. L., Horan, T. C., Gaynes, R. P., Pollock, D. A., & Cardo, D. M. (2007). Estimating health care-associated infections and deaths in US hospitals, 2002. Public health reports, 160-166.
- Joynson, J. A., Forbes, B., & Lambert, R. J. W. (2002). Adaptive resistance to benzalkonium chloride, amikacin and tobramycin: the effect on susceptibility to other antimicrobials. Journal of applied microbiology, 93(1), 96-107.
- Kampiatu, P., & Cozean, J. (2014). A controlled, crossover study of a persistent antiseptic to reduce hospital-acquired infection. African journal of infectious diseases, 9(1), 6-9.
- Center for Disease Control (2008) “Guideline for Disinfection and Sterilization in Healthcare Facilities”, Centers for Disease Control, 2008. Accessed October 19, 2016. http://www.cdc.gov/hicpac/pdf/guidelines/disinfection_nov_2008.pdf
- Seymour Stanton Block (2001). Disinfection, sterilization, and preservation (5th ed.). Lippincott Williams & Wilkins. p. 215. ISBN 0-683-30740-1.
- Gómez-López, V. M., Rajkovic, A., Ragaert, P., Smigic, N., & Devlieghere, F. (2009). Chlorine dioxide for minimally processed produce preservation: a review. Trends in Food Science & Technology, 20(1), 17-26.
- Cha, C. N., Lee, Y. E., Kang, I. J., Yoo, C. Y., An, S., Kim, S., & Lee, H. J. (2012). Bactericidal Efficacy of Vital-Oxide®, Disinfectant Solution Against Salmonella Typhimurium and Brucella Ovis. J Fd Hyg Safety, 27(1), 50-4.
- Stratilo, C. W., Crichton, M. K., & Sawyer, T. W. (2015). Decontamination Efficacy and Skin Toxicity of Two Decontaminants against Bacillus anthracis. PloS one, 10(9), e0138491.
- Pyrek, K. M. (2013). Clostridium difficile Prevention and Control Strategies. Infection Control Today. March. Accessed October 21, 2016. http://www.infectioncontroltoday.com/~/media/Files/Medical/Ebooks/2013/03/03-13ICT-CdiffReport-s.pdf
- Watt BE, Proudfoot AT, Vale JA. (2004) Hydrogen peroxide poisoning. Toxicol Rev. 23(1):51-7.
- Robertson JT (2016) Electrostatic Technology for Surface Disinfection in Healthcare Facilities. InfectionControl.tips. 10:1-4. https://infectioncontrol.tips/2016/10/14/electrostatic-in-healthcare/
- Wagenet L, Darling S, Lemley A (2004) Ultraviolet Radiation for Disinfecting Household Drinking Water Cornell Cooperative Extension, College of Human Ecology Fact Sheet 10, December 1993. Accessed October 21, 2016. http://waterquality.cce.cornell.edu/publications/CCEWQ-10-UVWaterTrtforDisinfection.pdf
- Cotton, C. A., Owen, D. M., Cline, G. C., & Brodeur, T. P. (2001). UV disinfection costs for inactivating Cryptosporidium. American Water Works Association Journal, 93(6), 82.












