Peer Reviewed
Conflicts of Interest
Barry Hunt – President & CEO of Class 1 Inc.
William Anderson – none.
Abstract
The use of mobile ultraviolet (UV) germicidal irradiation has been documented to reduce contamination levels and HAIs in hospitals. However, there is a general lack of information on the efficacy of wall-mounted, automated UV systems that are designed to irradiate rooms whenever they are left unoccupied. In this study, the Aseptix 1 UVC device (which includes two motion detectors, a magnetic door contact, and a microprocessor programmed to deliver a five-minute dose of UVC when the room is empty and the door closed) was evaluated. The number of contaminated surfaces decreased by approximately 65% overall and this was found to be a statistically significant result at a 95% confidence level. During periods when the UV systems were de-activated, the frequency of contaminated surfaces immediately reverted to the prior rate (within 24 hours). In addition, airborne contamination dropped by approximately 40%. However, there is less statistical confidence in this result due to fewer samples and more variability inherent in the settle plate method used.
Introduction
Environmental contamination in healthcare facilities leads to healthcare acquired infections (HAIs)1-7. The risk of acquiring an HAI increases from 39% to 353% when the prior room occupant has had an epidemiologically important HAI8-9. Mobile UV room disinfection at terminal discharge has been shown to be effective in reducing bacterial room contamination10-11 and in reducing the overall rate of HAIs12 – 13. UVC terminal room disinfection was recently shown to reduce HAIs of patients exposed to prior room occupants with epidemiologically important HAIs by 32% and 37% over terminal cleaning with quaternary ammonium compounds and bleach alone, respectively14.
While the impact of mobile UVC devices for surface disinfection have been well studied, fixed and automated UVC devices have not. One such device is the Sanuvox Aseptix 1, which is marketed for use in bathrooms, utility rooms, and equipment rooms in healthcare facilities. As Clostridium difficile and vancomycin-resistant enterococci (VRE) are intestinal bacteria characterized by diarrhea and there is evidence toilet aerosols contribute to environmental contamination and infections15 – 19, targeting bathrooms may have a significant impact on HAIs. Since utility rooms and equipment rooms form nexuses of staff and equipment traffic and interactions, targeting reductions in environmental contamination in these areas may also contribute to a reduction in HAIs. As such, the Aseptix 1 device warrants testing in real-world conditions. The goal of this work was to assess and quantify the impact of an Aseptix 1 automated UV disinfection system on surfaces and airborne microbial contaminant levels in hospital bathrooms, utility room, and equipment storage room.
Standard surface (contact plates) and air (settle plates) techniques were used to monitor the rooms on a five-day per week basis over a two-month period. The level of microbial contamination was monitored as a surrogate for potential pathogen contamination. The study was divided into three phases where two phases without UV disinfection were compared to a middle phase with the Aseptix 1 units active. A pair of patient bathrooms without UV disinfection was also monitored throughout the study as a control. All rooms were used normally, with no modifications to the routine cleaning and disinfection procedures already in place.
Study Purpose
To compare differences in microbial loads between rooms that have and do not have automated UV disinfection units installed and/or functioning. General microbial contamination levels were monitored to evaluate the overall efficacy of UV disinfection, and no attempt was made to monitor epidemiologically important pathogens or infection rates.
Study Design
Two patient room bathrooms (5-106A and 5-112A) were designated as “control” rooms, with no UV treatment applied during the study period.
Two patient room bathrooms (5-104A and 5-110A) were designated as the “test” rooms, with automated UV devices mounted on the walls in the area over the doors. Each bathroom had one Aseptix 1 device mounted over the door as per manufacturer’s instructions.
Two non-patient storage rooms (5-124A “Soiled Utility” and 7-116A “Equipment”) were also outfitted with UV devices. Manufacturer’s published UVC output data was used to generate UVC irradiance contour graphs in MathCAD based on dimensioned room drawings to establish the required number and optimal placement of Aseptix 1 devices to ensure room coverage. The utility room required three Aseptix 1 devices and the storage room required four. All devices within a room were wired in series with one device acting as a master device and the others as slave devices. Movement detected by any one of the multiple occupancy sensors would immediately interrupt the disinfection cycle as would the opening of the door.
Data loggers were installed to record second-by-second status of room occupancy, door opened / door closed, UV device on / off, date, and time.
The study was performed over the period of August 11 to October 16, 2015, with samples taken approximately daily from Monday to Friday of each week. The study was broken down into three periods:
- Period 1 – August 11 to September 8: no UV devices were active.
- Period 2 – September 9 to October 5: all UV devices were activated (excluding the “control” rooms).
- Period 3 – October 6 to October 16: no UV devices were active
The study design provides a continuous comparison with untreated (control) rooms (inter-room variability), as well as comparisons between pre- and post-UV testing for intra-room variability.
Normal housekeeping cleaning and disinfection practices were maintained in all rooms during the study periods.
The utility room and equipment room were each equipped with door closers, but not the patient bathrooms.
Signs were posted outside each room advising: “UV Testing in Progress. Please close the door.”
UV Devices
The UV devices installed were the Sanuvox Aseptix 1 automatic UVC room disinfection systems (Figure 1), supplied by Class 1 Inc. and installed by St. Mary’s General Hospital staff. The devices were programmed to provide 5 minutes of germicidal UV irradiation after a room was vacated and the door was closed, which was determined by door contacts and motion sensors. In the event that a closed room is not entered for 4 hours, the units are programmed to automatically provide 5 minutes of irradiation.

Figure 1. Aseptix 1 automated UVC room disinfection device that were wall-mounted in the bathrooms and storage rooms.
Sampling Locations
Within each of the study room, three surfaces were selected for routine microbial contamination sampling, and a “settle plate” was used as a qualitative indicator of airborne microbial contamination. Surfaces were selected in consultation with hospital staff and were expected to be areas with “high touch” frequency by patients, staff and visitors (for bathrooms), or only staff only (storage rooms). The suitability of these selected surfaces was confirmed in the first days of the study when high microbial counts were detected. The specific surface sample locations in each room are listed in Table 1 and example bathroom surfaces are illustrated in Figure 2. All rooms were used as normal, with no modifications to the routine cleaning and disinfection procedures already in place.
Table 1.: Sampling locations in each study room.
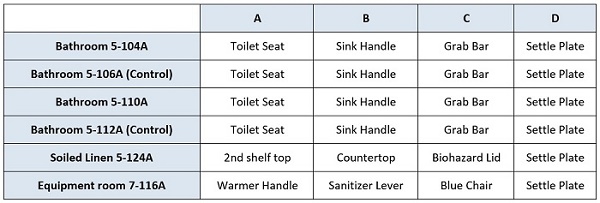

Figure 2. Examples of sampling locations in patient bathrooms: (top) toilet seat and grab bar to the left, and (bottom) sink handles.
Sampling Methodology
The sampling methodology was chosen based on standard practices in the pharmaceutical industry for monitoring microbial contamination in facilities [1, 2]. The sampling methodology is not specific to any particular microbe or pathogen, and was selected to assess the general extent of microbial contamination on surfaces and in the air. In this context, “microbial contamination” includes any bacteria, yeast, or fungi capable of growing at ~30°C on a non-selective solid medium (trypticase soy agar). Viruses are not detected with this sampling method. Surface sampling was performed using 65 mm diameter RODAC plates (replicate organism detection and counting; Figure 3) (VWR, Mississauga). Air sampling was performed by leaving 100 mm diameter Petri dishes (VWR, Mississauga) containing the trypticase soy agar (VWR, Mississauga) opened for a period of one-hour and then incubating the dishes at ~30°C for 72 hours. This “settle plate” method is qualitative because it does not sample a specific volume of air and is subjected to variations in air currents and room use.
Further details on the sampling materials and methods are given in the Appendix.

Figure 3. Example of a RODAC plate in use for sampling a surface (from www.biotestlabs.com)
Microbial Load Analysis Protocol
Settle plates and contact plates were incubated at 27–30°C for ~72 h. After incubation, colony counts were conducted and recorded, and photographs were taken to document the appearance and morphology of the colonies. Following standard practice, contact and settle plates that contained than 100 colonies after 72 h of incubation were considered “too numerous to count” (TNTC) and recorded as counts of 101. Plates that lacked distinct colonies due to heavy and overlapping growth were also considered TNTC.
Results and Discussion
UV Operations
Using the motion detectors and data loggers, the frequency of room use and disinfection were quantified during the latter part of the study period. These results are summarized in Table 2.
Table 2. Average number of times per day each room was entered, the number of UV disinfection events during the periods when the UV systems were activated, and the average fraction of entries where disinfection events were triggered. Averages are shown with ± one standard deviation, over the 18-day period when the UV systems were active.
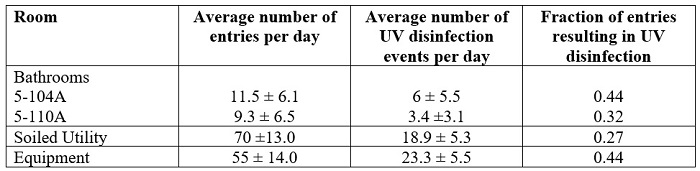
Table 2 demonstrates that the use of patient bathrooms was highly variable (as indicated by the large standard deviation relative to the average), which ranged from a minimum of 2 to a maximum of 30 entries. In contrast, the Soiled Utility and Equipment rooms were entered more frequently and consistently from day to day, with the number of entries ranging from 43 to 98. In addition, also it was observed that not every entry resulted in a triggered UV disinfection event, likely due to insufficient time between entries to complete a disinfection cycle and the lack of door closures. In general, ~30–45% of entries resulted in UV disinfection. However, the fraction is lower for the Soiled utility room, possibly due to staff propping the door open for lengthy periods to facilitate movement of carts, mop buckets, and equipment. The patient bathrooms did not have door closing mechanisms. Patient and staff compliance with closing the bathroom doors (32–44%) was similar to that of the equipment room (44%), which did have a door closing mechanism. All rooms could benefit with more frequent door closings.
Surface Disinfection
The study was conducted in three distinct phases. In the first phase (18 days) to provide background data, the rooms were sampled without any active UV disinfection. In phase 2 (18 days), the UV systems were activated in the two test patient bathrooms and two other rooms (Soiled Utility and Equipment), while two other patient bathrooms with no UV were sampled to provide ongoing “control” data. In phase 3 (9 days), all the UV devices were de-activated and sampling continued in all the rooms.
Sampling generally took place in the late morning or by mid-afternoon for all locations. Rooms without active UV disinfection consistently showed TNTC results in 40–60% of the surfaces sampled. Figure 3 shows an example of a TNTC plate.
Figure 4. A typical TNTC (too numerous to count) result from October 14th (room 5-104A toilet seat), showing a large number of distinct small colonies due to heavy contamination.
The contact plates generally showed one of two extremes, either TNTC or colony counts in the order of 5–20. Therefore, we decided to compare the number of TNTC results for each room during the different study phases for data analysis. To normalize the TNTC results for a consistent comparison, the number of TNTC results for each room (all three sampled surfaces combined) were divided by the number of days in each phase and then multiplied by 5 days. This resulted in a normalized number of TNTC counts per 5-day period for each room. For the patient bathrooms, the results are summarized in Figure 5.
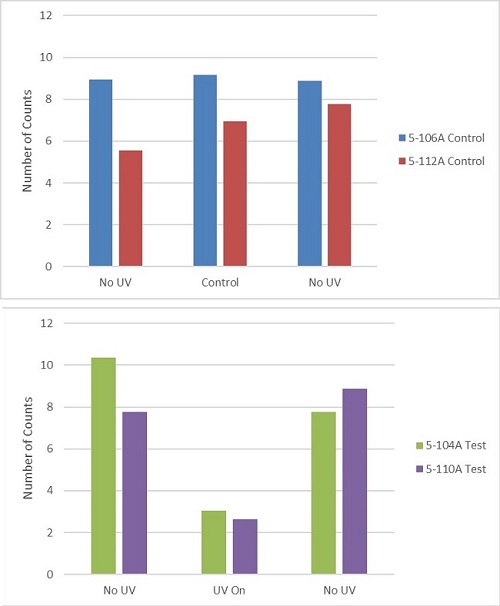
Figure 5. Number of TNTC counts (normalized to a 5-day period) for each patient bathroom over the three phases of the study. Top: counts for the “control” rooms, where no UV was active during any phase. Bottom: counts before, during, and after the automated UV activation in the two test rooms.
The counts in the “control” rooms were relatively consistent throughout the study, ranging from ~6–9 TNTCs per 5-day period (Figure 5). Statistical significance testing using the Poisson distribution indicated that these variations are within normal expectations for random sampling (i.e. the differences between the control rooms over the 3 phases were not statistically significant).
For the “test” rooms, when the UV systems were activated in the 2nd phase (UV On), the TNTCs per 5-day period dropped to ~3. Comparing all the control and test TNTCs together, a 65% decrease in heavy microbial contamination was achieved when the UV systems were activated. This change was found to be statistically significant at a 95% confidence level.
Notably, when the UV systems were subsequently switched off again, the TNTCs rapidly rose back to their original levels, within expected statistical variation. A review of the underlying data indicates that this elevation in TNTCs occurred within 24–48 h of the UV lamps being switched off, suggesting that recontamination or re-growth on these touch surfaces is a relatively rapid process.
Using a similar approach, the results for the utility and equipment rooms are summarized in Figure 6. In both rooms, the TNTCs dropped once the automated UV system was activated, and this drop was statistically significant for both rooms at a 95% confidence level. During the UV activation phase, the incidence of TNTC counts decreased by 61% and 83% for the Soiled Utility and Equipment rooms, respectively.
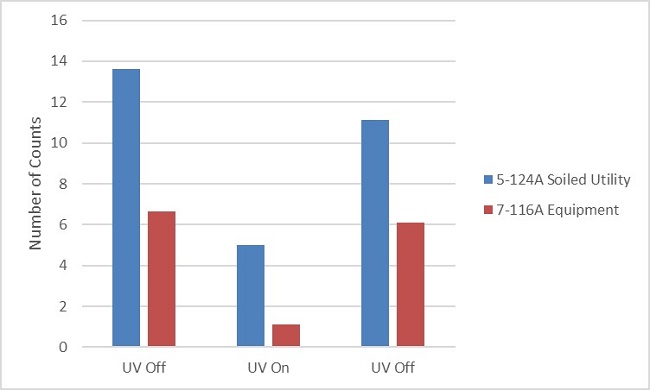
Figure 6. The number of TNTC counts (normalized to a 5-day period) for each non-patient storage room over the three phases of the study.
Overall Comments on Treatment Efficacy
When all the data are pooled and considered together, the activation of the automated UV treatment system resulted in a 65% decrease in heavy microbial contamination of surfaces. This decrease was consistent across rooms and was statistically significant at a high 95% confidence level. For the types of rooms and surfaces studied in this work, it can be concluded that the automated UV system successfully reduces microbial contamination.
UV Effects on Airborne Microbial Loadings
The settle plate results provided an indication of airborne microbial loadings. However, they were not as rigorous and quantitative as surface sampling because there was no control of air flow patterns and volumes of air that may have brought microbes into contact with the plate surfaces. These plates are also susceptible to random contamination events during the one-hour exposure period, such as coughing or sneezing by people in the vicinity of the plates, or toilet flushing which may generate aerosols. However, they do provide some measure of general airborne contamination of potential interest.
Figure 7 shows the colony counts on the settle plate samples from each patient bathroom (one per room daily) over the entire study period. In the control rooms with no UV (Figure 7 top), it can be observed that the airborne contamination varied, but TNTC counts were frequently occurring throughout the study. In contrast, for UV test rooms (Figure 7 bottom) when the UV lights were activated, there is an apparent 40% reduction in counts and frequency of TNTC results. When the UV lights were subsequently de-activated, the frequency of TNTC results appeared to rapidly increase. As shown in Figure 7, the reduced frequency of high colony counts during the UV activation period supports the theory that the systems maintained lower airborne microbial loadings. However, the apparent differences are not strongly supported by statistical significance tests (probability = 0.31). This could be due to a limited number of data points (only one sample per room per day) or a lack of sensitivity in the test method. Despite the lack of statistical rigor, the results are directionally what would be expected.
Figure 8 shows the settle plate results for the two non-patient storage rooms. During the phase with UV lights activated (horizontal arrow), the apparent airborne contamination appeared to decrease substantially. The low colony counts and sample numbers make rigorous statistical testing inconclusive, but the results do not contradict the expectations. There are noticeable differences in contamination loading between the two rooms with the Soiled Utility room showing much more frequent TNTC results. This may be due to differences in the nature of the materials being brought into the two rooms and the way the materials were handled. Some activities in the Soiled Utility room appeared to generate airborne contamination much more readily than activities in the Equipment room. In addition, it can be observed that airborne contamination in the patient bathrooms was consistently much higher than in the non-patient storage rooms (Figure 7 and 8). This is presumably due to patient coughing, sneezing, and toilet/sink use in the more confined areas, which may be detected more readily by the settle plate method.
Overall, although the settle plate method for airborne contamination measurement is not highly quantitative and is subject to various sampling errors, it does appear to support the observation that the automated UV disinfection system assists with reducing airborne microbial contamination in the test rooms.
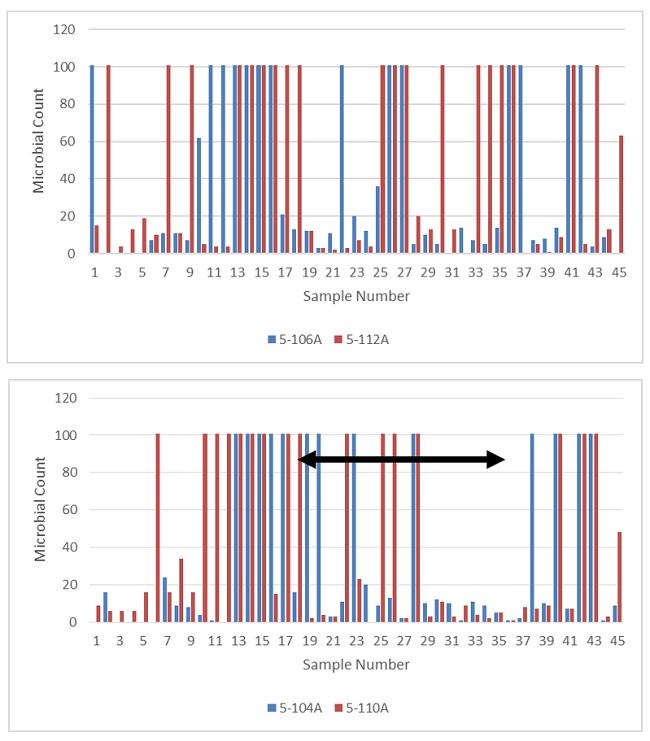
Figure 7. Settle plate (airborne) colony counts in the patient bathrooms. TNTC results are coded as a colony count of 101. Each room had one daily settle plate sample, exposed for ~1 h. Top: results from the two “control” rooms without UV disinfection. Bottom: results from the two test rooms. The horizontal arrow indicates the time period where the automated UV lights were active (samples 19 to 36), with a 40% decrease in TNTC frequency.
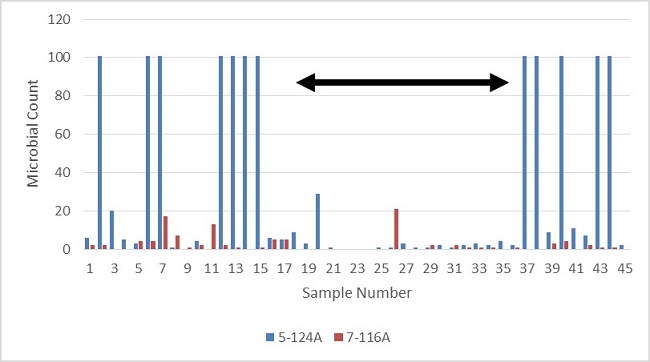
Figure 8. Settle plate (airborne) colony results in the Soiled Utility (5-124A) and Equipment (7-116A) rooms. TNTC results are coded as counts of 101. The horizontal arrow indicates the time period where the automated UV lights were active (samples 19 to 36).
Irradiance Measurements and Predicted Disinfection
Although the experimental measurements support the observation that the automated UV system was effective at reducing surface and airborne microbial contamination, it is useful to corroborate this work with predictions based on UV disinfection theory and literature. Therefore in this section, the predicted level of disinfection is determined for a 5-minute UV cycle.
The actual UV irradiance was measured at several sampling locations in each test room to quantify how much 254 nm UV dose was being delivered to a surface during a 5-minute disinfection event. The measurement locations were chosen to correspond approximately to those locations sampled for microbial load. However, it was not feasible to measure at the grab-bar location in the patient bathrooms due to the geometry of the UV sensor. These results are summarized in Table 3.
Table 3. UV irradiance measurements at various sampling locations in each test room and the corresponding UV dose for a 5-minute cycle.
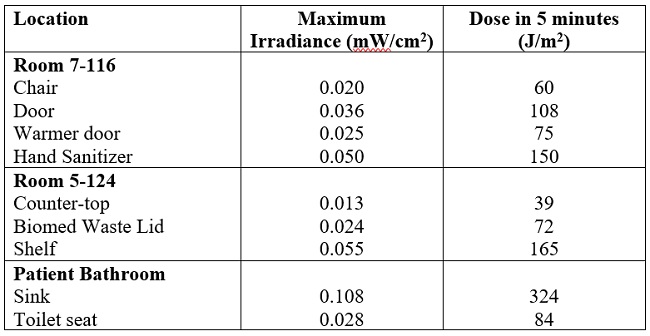
The UV measurements in Table 3 appear to be consistent with expected trends, where the irradiance should be higher in locations that are closer to the UV device(s). For example, since the UV device, which was mounted above the door in the patient bathroom, was closer to the sink than the toilet seat, the sink had a higher irradiance.
The measured doses in Table 3 can be used with published data (Tables 4 and 5) to estimate the expected level of disinfection that occurs during each 5-minute disinfection event when the UV lights automatically turn on. For this estimation, the following standard equation is used:
Reduction (%) = 100(1-exp(-kD))
or
Log Reduction = kD/2.303
where k is the “rate constant” for a specific microbe (Table 4) or a class of microbes (Table 5), and D is the delivered UV dose.
Table 4. Reported UV rate constants (k) for selected microbes on surfaces, from Kowalski [3, 7]. The rate constant for C. difficile is based on C. perfringens [7].
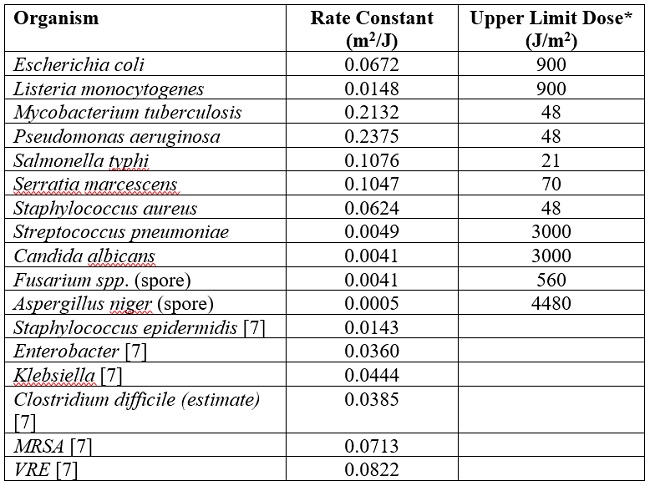
* highest dose for which the rate constant was measured. Extrapolation above this limit is uncertain. The rate constants are for a first order disinfection model of the form: S = exp(-kD), where S is survival fraction, k is the rate constant, and D is the dose (assuming a single stage decay response with no threshold or shoulder).
Table 5: Average rate constants (k) for various types of microbes, adapted from Kowalski [3] (RH = Relative Humidity).
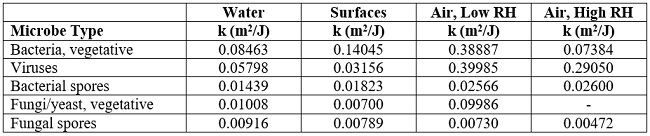
Table 6: Estimated reductions in microbial load of selected types of organisms for a 5-minute dose of UV in the test rooms, using the data given in Table 3 and generic surface disinfection kinetics given in Table 5.
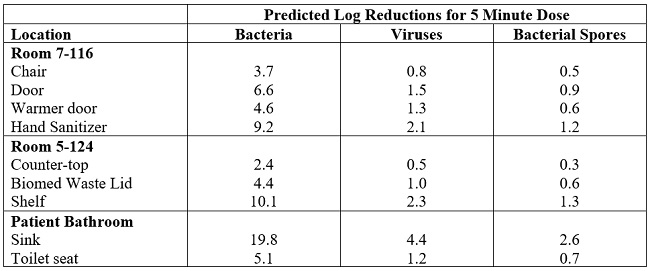
Estimated reductions for generic bacteria, viruses, and bacterial spores are given in Table 6, based on the average rate constants given in Table 5. It can be observed that bacteria on surfaces in a patient bathroom are predicted to be significantly reduced (Table 6). For example, by more than 5 log (i.e. 99.99%) on the surface of a toilet seat. Bacterial reductions on surfaces in the other rooms are predicted to range from 2 log (99%) to 10 log (>99.99%), depending on the location and distance from the UV devices. Due to their higher resistance to UV disinfection, predicted reductions are significantly lower for viruses and bacterial spores. Nevertheless, disinfection levels approaching or exceeding 90% are achievable in most locations, even for these more resistant classes of organisms.
There is no reliable way to compare these UV dose estimates with the measured microbial counts shown in Figures 5 and 6, due to a lack of knowledge of initial microbial loads, the frequency and nature of the room use, and the amount of added microbial contamination with each use. However, the UV dose estimates provide a theoretical justification for concluding that the UV effect is real and significant, as supported by the data in Figures 5 and 6.
The results in Table 6 also suggest that the 5 minute UV cycle time is reasonable for the control of bacterial contamination. However, if more stringent control of viruses and spores is desired, an increase in cycle time could be considered.
For pathogens of particular interest, predicted levels of disinfection can be calculated using the published rate constants given in Table 4, and assuming a UV dose of 120 J/m2 (the average of measurements from this study; Table 3). Predictions are given in Table 7.
Table 7: Predicted disinfection levels for selected pathogens, for a 5-minute cycle with an average UV dose of 120 J/m2.
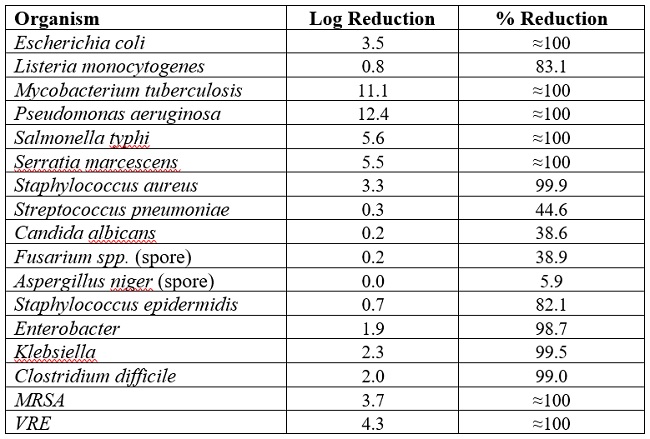
The estimates in Table 7 suggest that the UV systems are capable of >80% microbial disinfection for most bacteria, with exceptions that include fungi or microbes in spore form. Since susceptibility to UV disinfection can vary with microbial strains, species, and with environmental conditions, these results should not be taken as precise predictions. However, they are useful indicators of general levels of disinfection.
For air disinfection, it can be noted in Table 5 that the rate constants in the air at high relative humidity (Hi RH) are lower for bacteria and higher for viruses than the respective rate constants for surfaces. Therefore, it might be expected that the UV devices will be more effective at disinfecting airborne viruses and somewhat less effective for airborne bacteria.
Conclusions
Surface sampling in four patient bathrooms, an equipment storage room, and a soiled utility room showed that the use of Sanuvox Aseptix 1 automated UV disinfection devices significantly reduced microbial contamination. The frequency of heavily contaminated surfaces, characterized by TNTC samples, was reduced by ~65% overall during the periods when the UV systems were active and door closings ranged from 27– 44%, compared to periods when the UV systems were not active.
Airborne contamination, as detected by simple “settle plate” samples, was also reduced by ~40%. However, the statistical significance of these results was not as strong due to a limited sample number.
Further study is required to determine if implementation of this new paradigm of fixed, automated, targeted, UVC disinfection in hospital bathrooms, utility rooms, and equipment rooms leads to significant reductions in HAIs.
References:
- Weber, Kanamori, Rutala. Environmental Contamination Leads to Healthcare Acquired Infections. Curr Op Infect Dis .2016. In press.
- Weber DJ, Rutala WA, Miller MB, Huslage K, Sickbert-Bennett E. Role of hospital surfaces in the transmission of emerging health care-associated pathogens: Norovirus, Clostridium difficile, and Acinetobacter Am J Infect Control 2010;38:S25-33.
- Boyce JM. Environmental contamination makes an important contribution to hospital infection. J Hosp Infect 2007;65(Suppl 2):50-4.
- Rutala WA, Weber DJ. The role of the environment in transmission of Clostridium difficile infection in health care facilities. Infect Control Hosp Epidemiol 2011;32:207-9.
- Hota B. Contamination, disinfection, and cross-colonization: are hospital surfaces reservoirs for nosocomial infection? Clin Infect Dis 2004;39:1182-9.
- Dancer SJ. Importance of the environment in methicillin-resistant Staphylococcus aureus acquisition: the case for hospital cleaning. Lancet Infect Dis 2008;8:101-13.
- Otter JA, Yezli S, French GL. The role played by contaminated surfaces in the transmission of nosocomial pathogens. Infect Control Hosp Epidemiol 2011;32: 687-99.
- Huang S, Datta R, Platt R. Risk of acquiring antibiotic-resistant bacteria from prior room occupants. Arch Intern Med 2006;166:1945-51.
- Rutala, W. Disinfection and Sterilization: The Good, The Bad, and The Ugly. Plenary presentation, American Professional Infection Control annual conference, 2016
- Boyce J, Havilla N, Moore B. Terminal Decontamination of Patient Rooms Using an Automated Mobile UV Light Unit Infect Control Hosp Epidemiol 2011:32: 737-742
- Rutala WA, Gergen MF, Weber DJ. Room decontamination with UV radiation. Infect Control Hosp Epidemiol 2010;31:1025-9.
- Haas J, Menz J, Dusza S, Montecalvo M. Implementation and impact of ultraviolet environmental disinfection in an acute care setting. American Journal of Infection Control 42 (2014) 586-90
- Anderson DJ, Gergen MF, Smathers E, Sexton DJ, Chen LF, Weber DJ, et al. Decontamination of targeted pathogens from patient rooms using an automated ultraviolet-C-emitting device. Infect Control Hosp Epidemiol 2013;34: 466-71.
- Andersen, DJ, et al. The Benefits of Enhanced Terminal Room (BETR) Disinfection Study: A Cluster Randomized, Multicenter Crossover Study with 2×2 Factorial Design to Evaluate the Impact of Enhanced Terminal Room Disinfection on Acquisition and Infection Caused by Multidrug-Resistant Organisms (MDRO). SHEA Featured Abstract Society of Healthcare Epidemiology of America, IDWeek October 9, 2015, San Diego, California.
- Barker J., Jones MV. The potential spread of infection caused by aerosol contamination of surfaces after flushing a domestic toilet. Journal of Applied Microbiology 2005:99: 339–347
- Best E., Fawley W., Parnell P, Wilcox M. The Potential for Airborne Dispersal of Clostridium difficile from Symptomatic Patients. Clinical Infectious Diseases 2010; 50(11):1450–1457
- Johnson DL, Mead KR, Lynch RA, Hirst DVL. Lifting the lid on toilet plume aerosol: A literature review with suggestions for future research. American Journal of Infection Control 2013: 41: 254 – 258
- Roberts K, Smith C, Snelling AM, Kerr K, Banfield KR, Sleigh PA, Beggs CB. Aerial Dissemination of Clostridium difficile spores BMC Infectious Diseases 2008: 8: 7
- Verani M, Bigazzi R, Carducci A. Viral contamination of aerosol and surfaces through toilet use in health care and other settings. American Journal of Infection Control: 2014: 42: 758-62
Acknowledgements
The assistance of Laura Copeman (Class 1 Inc.), Roger Holliss, Kelly Metcalfe, Gerald LeMoine, Chris Hope (St. Mary’s General Hospital), and Shazia Tanvir (University of Waterloo) with the set up and operation of the study is greatly appreciated and acknowledged.
Appendix
Sampling Materials
- Trypticase Soy Agar with Lecithin and Polysorbate 80
- Sterile Petri dishes: 100 mm and 65 mm diameter
- “Contact plates” consisting of sterilized agar in the 65 mm Petri dishes, also referred to as RODAC plates (replicate organism detection and counting; Figure 1). Store in refrigerator, do not freeze, and minimize exposure to light. Store with agar surface up and lid down to minimize contamination from condensation.
- “Settle plates” consisting of sterilized agar in 100 mm Petri dishes
- ATP swabs (e.g. Hygiena ultrasnap) and ATP meter
Sampling Protocol: at St. Mary’s General Hospital (SMH)
- Sampling at SMH was performed by Class 1 (Laura Copeman)
- For each day, review with SMH staff and acquire PPE (if needed)
- For each sample location, record date, time, time when last cleaned, type of cleaning, and any other relevant details
- For each room:
- Wear gloves during handling of contact plates to minimize contamination from hands.
- Apply contact plate to sample locations in Table 1. Label the plate and tape the lid to prevent accidental opening. Wipe clean the toilet handle to remove any residual agar.
- Swab one location in each room with the ATP test swab (in a spot not touched by the RODAC plate). Take reading as directed and record. Wipe clean the sample location.
- Place settle plate in the same place each time and remove cover. Leave plate for 1 h, cover the plate with the lid, and remove from location. Label the plate.
Repeat the protocol and use the same sampling locations with each subsequent visit, if possible. Note any deviations from the protocol or changes in sample locations.













[…] bathrooms to control the background bacterial contamination between uses. The paper can be read on this website. The data indicated that it was possible to dramatically lower bacterial contamination levels […]