Peer Reviewed
Disclosure statement: The authors of this report and InfectionControl.tips declare no conflict of interest with the following critical evaluation and research. No funds or influence were provided to InfectionControl.tips by any parties.
Abstract
Hospital wash basins are well known and established sources of hospital-acquired infections (HAIs)–contributing to increases in morbidity, mortality, prolonged hospital stay, and cost. Therefore, there is a need for interventions that can reduce HAIs by eliminating sources of contaminants or reservoirs for pathogens. Even more worrisome, vancomycin-resistant Enterococci (VRE) and MRSA are most commonly identified in wash basins. In this study, we investigated the efficacy of a disposable wash basin liner to reduce HAIs. The Basin Glove disposable liner was shown to be effective at reducing cross contamination and thus, potentially eliminating a possible source of HAIs in the hospital.
Introduction
Hospital-acquired infections (HAIs) have been widely studied and vast amounts of research have been conducted to reduce and prevent the spread of such infections.1 Despite great effort to reduce and prevent HAIs, the CDC published the HAI Prevalence Survey which reported an estimated 722,000 HAIs in the United States in acute care hospitals with an associated 75,000 deaths during hospitalization. In addition, more than 50% of HAIs occur outside the intensive care unit.2 The American Journal of Infection Control indicated HAIs are the most common complication among hospitalized patients and are among the top 10 leading causes of death in the United States.3 Many studies have been conducted to identify possible reservoir sources associated with HAIs, which include used bath water in hand washing sinks, biofilm formation in water sources, cross contamination between patients due to poor hygiene, and soiled bath basins left out in the patient room.3,4 Similarly, the American Journal of Infection Control reported many contributing sources of HAIs that are common place in the hospital setting–biofilms formed in water pipes, hot water tanks, air condition cooling towers, and bathing basins.4 Many infection control efforts have been implemented to prevent and reduce such outbreaks from occurring, including water chlorination, filtration, thermal disinfections, and UV irradiation.4 Despite the great effort to reduce HAIs, the CDC published a report in 2017 indicating there has been little to no decline in HAIs, including methicillin-resistant Staphylococcus aureus (MRSA) infections. 5
Many studies have reported that bathing basins are a major HAI reservoir and has not been addressed sufficiently to reduce the spread of infections.3,4 A study including some 88 hospitals found that about 2/3 of bath basins harbored at least one pathogen associated with HAI, with vancomycin-resistant Enterococci (VRE) and MRSA as the most commonly identified.3 Wash basins are used for incontinence clean up, indwelling catheter care, emesis collection, and common bathing.3 In addition, the study reported that basins are often not replaced during the entire patient stay, with an average stay of 8.1 days, and the basins typically are being reused. Most patients in the hospital setting do not have shower privileges and basins have become standard use in acute and intensive care, and most care departments in the hospital.4 During bathing, skin flora may contaminate the water in the basins which may then spread via inhalation, ingestion, or with direct contact with compromised skin. The report confirmed that potential harmful microorganisms are present in bath basins even after the water is removed. There is a great need for novel intervention to prevent HAI associated with bath basins.4 The purpose of this study was to evaluate the efficacy of Basin Glove disposable wash basin liner in the reduction and prevention of possible pathogens associated with nosocomial infections and outbreaks. The Basin Glove disposable wash basin liner is a physical barrier used to cover the common wash basin and then be discarded after use.
Materials and Methods
Bacteria Growth
Bacterial cultures (Escherichia coli ATCC 11229 and MRSA ATCC BAA-44) and media were obtained from Hardy Diagnostics (Santa Monica, CA). MRSA or E. coli cultures were incubated for 18 hours at 37oC with continuous rocking in 300 mL tryptic soy broth (TSB) to a density of 1´109 CFU/mL.
Basin Treatment
Thirty standard wash basins were used. Each basin was labeled and marked for identification and separated into 3 groups (A, B, and C). A 4 cm2 area was outlined in each basin to control swabbing sample size. Following incubation, 300 mL sterile 100% glycerin was added to each culture to augment the viscosity of normal body fluids (blood, feces, or emesis), which have been commonly associated as contamination sources of wash basins. Group A basins were lined with a Basin Glove disposable liner and exposed to 100 mL culture+glycerin mixture by pouring the mixture into the wash basin for 30 seconds then transferring it to the next wash basin, repeating through all 10 basins. This volume and timing allowed the entire area of the basin to be covered by the bacterial solution. The liner was removed and discarded. The procedure was repeated with Group B basins, but without the Basin Glove disposable liner. Following the 30-second incubation with the bacterial mixture, the Group B basins were rinsed vigorously with tap water for 30 seconds (a common practice conducted in the hospital setting). In contrast, following the 30-second incubation period, no rinse was performed for Group C basins. This group was used to mimic the common practice of pouring out water from wash basin after use without a subsequent rinse. For each group, a cotton swab was dipped into sterile water and then the 4 cm2 area was swabbed in each basin. The swab tip was cut off and placed into a 5 mL TSB tube. Each tube was vortexed for 1 min, 10-fold serially diluted, and plated on tryptic soy agar (TSA) plates. The plates were incubated at 37oC for 24 hours and the colonies were counted.
Figure 1. Typical wash basin with and without Basin Glove liner.

Results
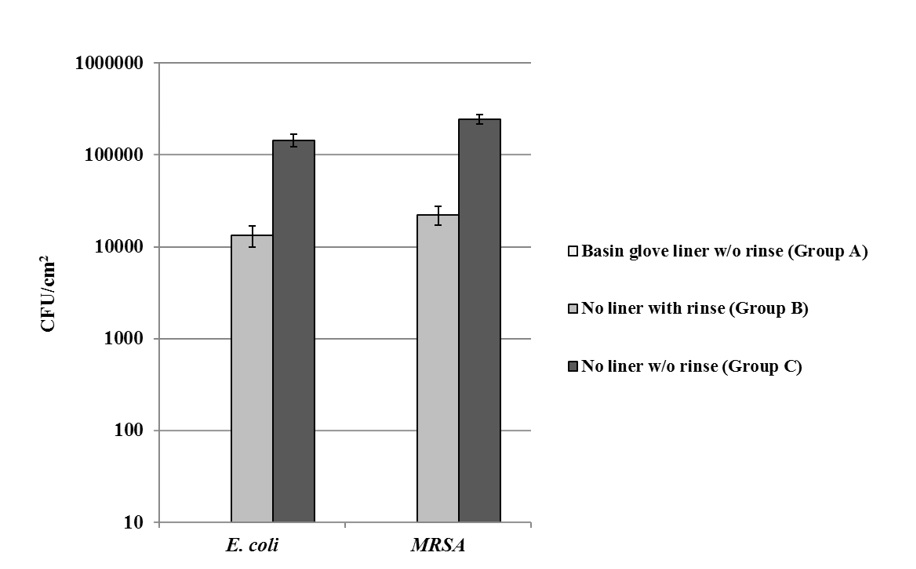
The purpose of this study was to evaluate the level of residual bacteria left in the wash basins following exposure to bacteria and a) not rinsing, b) rinsing with tap water, or c) lining the wash basin with a Basin Glove liner. An example of a typical wash basin with and without the Basin Glove liner is shown in Figure 1. Under our conditions, when the water basins were exposed to E. coli or MRSA without a rinse, there was over 100,000 CFU/cm2 (Fig. 2). When the wash basins were exposed to the bacteria and then rinsed with tap water, approximately 20,000 CFU/cm2 were present (Fig. 2). No growth was detected when the wash basins were covered with the disposable Basin Glove liner prior to bacterial exposure; minimum detection limit of 10 CFU/cm2 (Fig. 2).
Figure 2. Reduction in CFU/cm2 with wash basins lined with Basin Glove liner.
Wash basins with or without Basin Glove liner were exposed to E. coli or MRSA. Basins with no liner were either not rinsed or rinsed with tap water. Bacterial CFU/cm2 in each basin after treatment is shown. Values shown with error bars represent the mean +/- SEM of 10 replicate samples for each treatment. A paired t-test of Group A vs. Group B or Group C resulted p<0.00001 for both bacterial species.
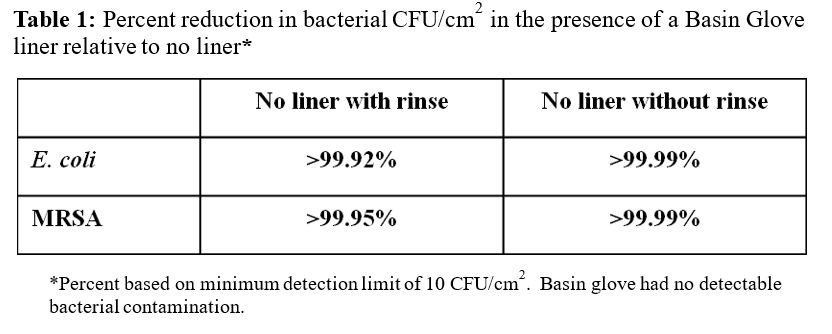
Each group was replicated 10 times for validation and calculation of standard deviations. There was a reduction (>99.9%) in bacterial contamination when the wash basin was lined with Basin Glove compared to when the wash basin was exposed to E. coli or MRSA without a rinse (based on a detection limit of 10 CFU/cm2; Table 1). In addition, there was a reduction in E. coli and MRSA (>99.92% and >99.95%, respectively) contamination in wash basins lined with Basin Glove compared to wash basin exposed to bacteria and then rinsed with tap water (based on a detection limit of 10 CFU/cm2; Table 1).
Discussion
This study tested the efficacy of the Basin Glove disposable liners as a possible tool to prevent or reduce community acquired pathogens associated with HAIs. The results demonstrated that the Basin Glove disposable liner is effective at reducing bacterial contamination on wash basins as there was no detectable bacterial contamination after use (based upon minimum detection limit of 10 CFU/cm2). A greater than 99.92% reduction in E. coli and MRSA contamination was observed with the use of the Basin Glove disposable liner compared to wash basins without a liner but were briefly rinsed. Similarly, >99.99% reduction in E. coli and MRSA contamination with using the Basin Glove disposable liner compared to wash basins without a liner and were not rinsed. As hypothesized, washing of the basin with tap water reduced the level of bacterial contamination approximately 5-fold (Fig. 2). However, there was a significant level of bacteria present in the basin, which in a hospital environment could lead to HAIs. Only the presence of the wash basin liner led to bacterial loads being reduced to a non-detectable level.
HAIs have been attributed to rising hospital costs due to increases in patient morbidity and mortality. Many studies have indicated that water basins as a common and major reservoir for microorganisms associated with HAIs. Our results suggest that the Basin Glove disposable liners can be used to reduce bacterial cross contamination related to water basins and thus, potentially preventing the transmission of HAIs.
References
- Anaissie, E. J., Penzak, S. R. & Dignani, M. C. The Hospital Water Supply as a Source of Nosocomial Infections. Arch. Intern. Med. 162, 1483 (2002).
- Centers for Disease Control and Prevention (CDC). HAI Data and Statistics. (2018). Available at: https://www.cdc.gov/hai/surveillance/index.html
- Marchaim, D. et al. Hospital bath basins are frequently contaminated with multidrug-resistant human pathogens. Am. J. Infect. Control 40, 562–564 (2012).
- Johnson, D., Lineweaver, L. & Maze, L. M. Patients’ bath basins as potential sources of infection: A multicenter sampling study. Am. J. Crit. Care 18, 31–38 (2009).
- Centers for Disease Control and Prevention (CDC). Healthcare-associated Infections in the United States, 2006-2016: A Story of Progress. (2018). Available at: https://www.cdc.gov/hai/surveillance/data-reports/data-summary-assessing-progress.html













[…] Download Image More @ infectioncontrol.tips […]